PDF) Statistical Considerations on Subgroup Analysis: Interpretation of clinical trial findings and study design for targeted subgroup

Anovus - While students of most universities are idle during the lock-down, Anovus students are moving ahead with accelerated speed, gaining additional qualifications from world-class universities like John Hopkins University, University of

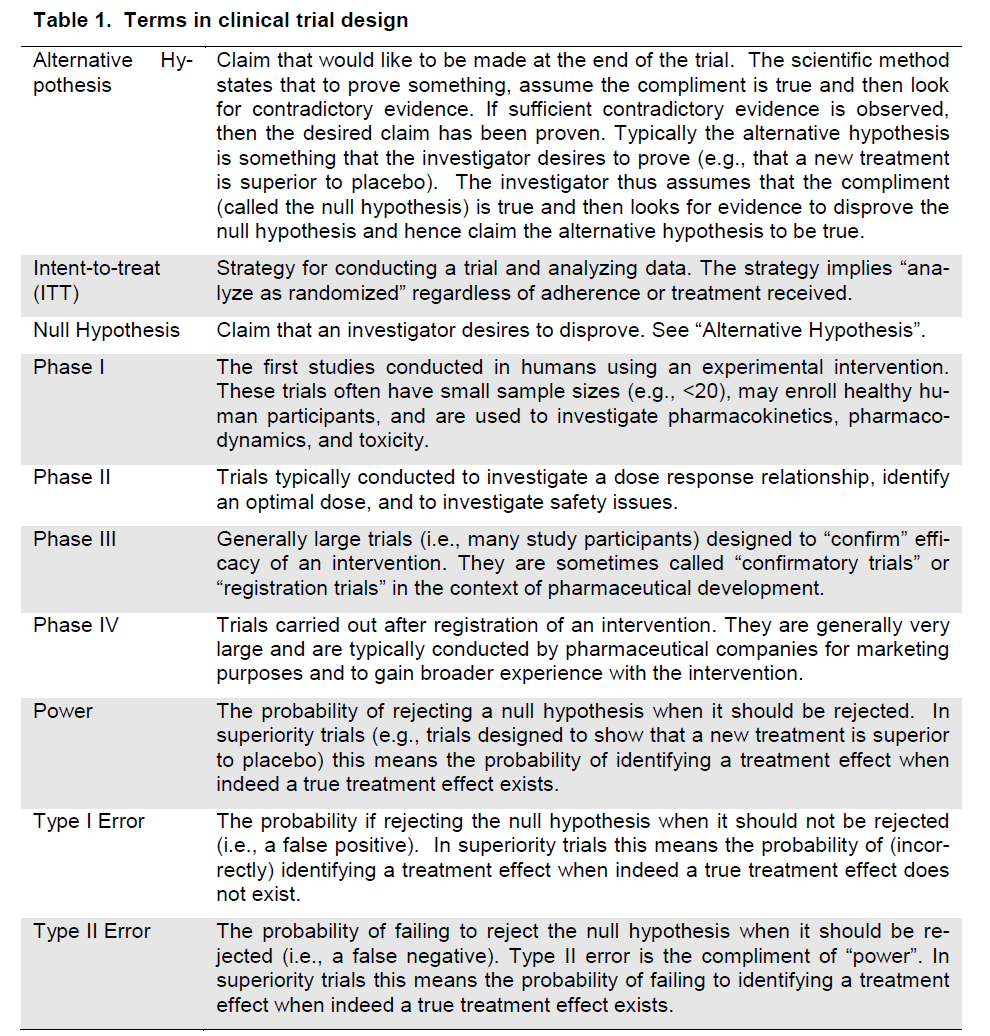
Figure 3. | A Primer on the Design, Conduct, and Interpretation of Clinical Trials | American Society of Nephrology

PDF) Revisiting the Design of Phase III Clinical Trials of Antimalarial Drugs for Uncomplicated Plasmodium falciparum Malaria

Should Clinical Trial Interpretation Be Dominated by A P Value of 0.05 for the Primary Endpoint? | tctmd.com

Medical Device Clinical Trial Design, Conduct, And Analysis | IEEE Conference Publication | IEEE Xplore
Bayesian methods for the design and interpretation of clinical trials in very rare diseases - Lancaster EPrints

Education and Training, icddr,b - Course Title: Introductory Course on Design and Interpretation of Clinical Trial Course Details: http://training.icddrb.org/siteinfo/circularDetail?1=1&view=300 Duration: 17, 18 & 25 February 2021 (3 Days) Last date of ...

Free online course: 'Design and Interpretation of Clinical Trials' – The Publication Plan for everyone interested in medical writing, the development of medical publications, and publication planning
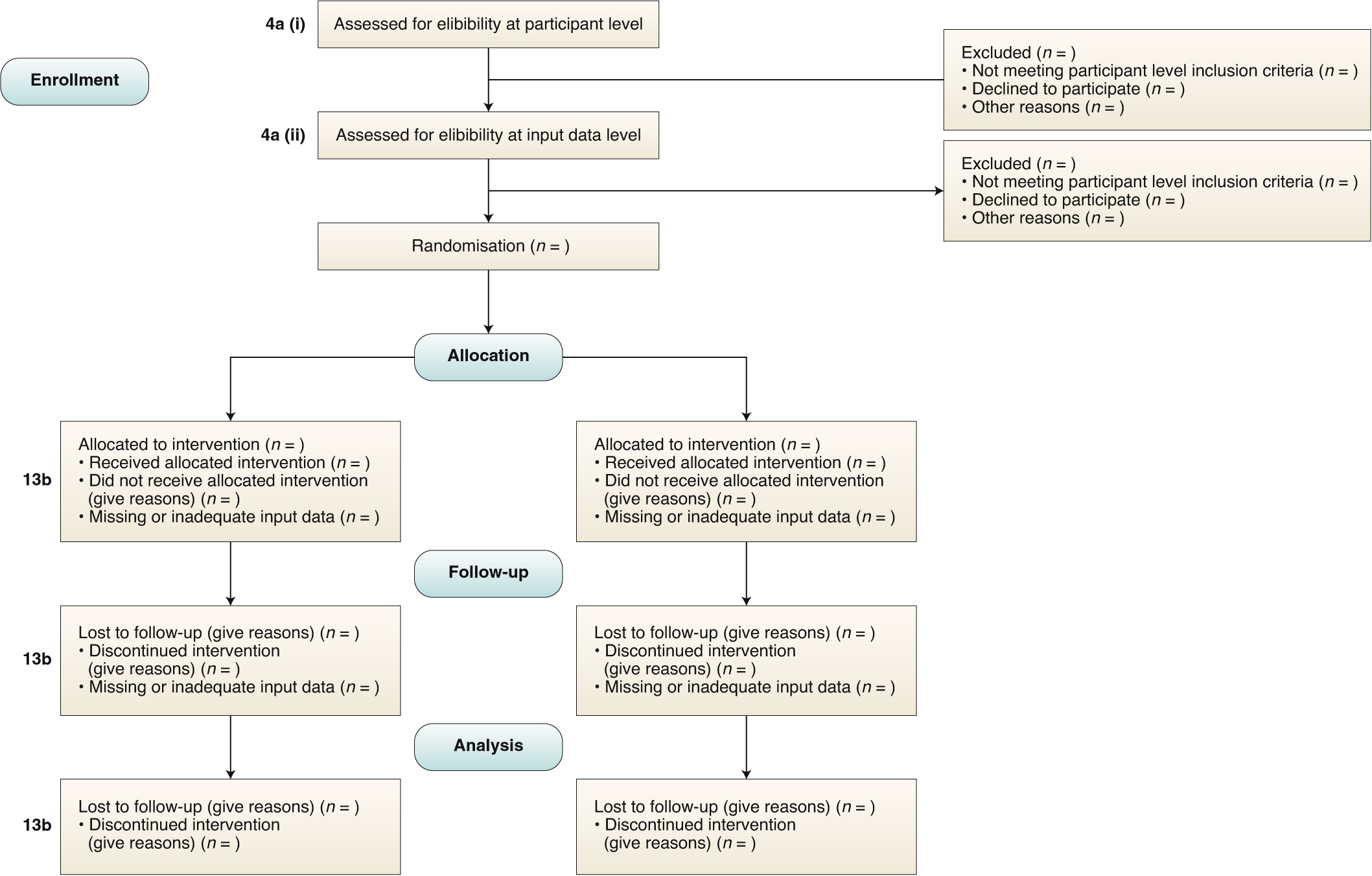
Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension | Nature Medicine

A Primer on the Design, Conduct, and Interpretation of Clinical Trials | American Society of Nephrology

4th Global Clinical Trial Advanced and Planning Symposium that will be scheduled on 27th February has been cancelled. <br>"4th Global Clinical Trial Advanced and Planning Symposium~ULTIMATEBILITY"<br>|Global Clinical Research Support Group